A02:Role of iron metabolism in immune cells in the pathogenesis of NASH
Takayoshi SUGANAMI
(Research Institute of Environmental Medicine, Nagoya University)
Lab website

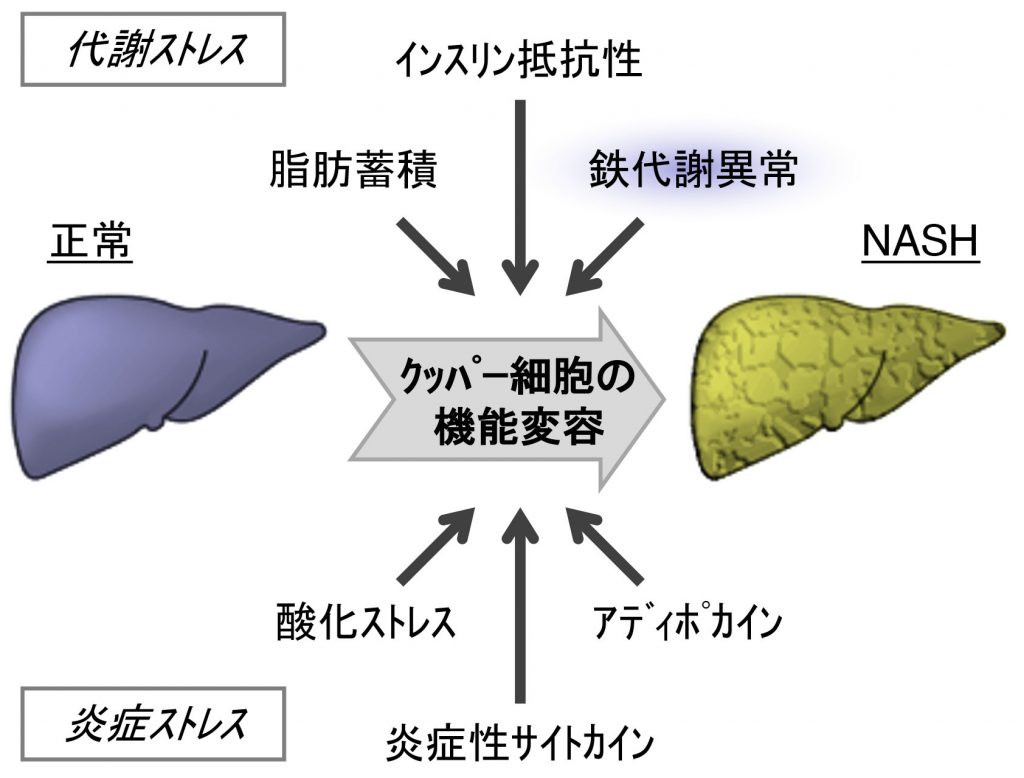
Dysregulation of iron homeostasis is frequently observed in patients with nonalcoholic steatohepatitis (NASH), which is characterized by steatosis with lobular inflammation and fibrosis. However, it remains unclear how disrupted iron metabolism contributes to the pathogenesis of NASH. Herein, we will investigate the role of macrophage iron metabolism in the pathogenesis of NASH using genetically obese melanocortin 4 receptor-deficient (MC4R-KO) mice that develop NASH on Western diet. Hepatic macrophages are separated into ferromagnetic (Fe-hi) and non-ferromagnetic (Fe-lo) populations via magnetic column separation. Our preliminary data showed that Fe-hi macrophages isolated from the livers of the NASH model exhibit increased expressions of proinflammatory and profibrotic factors. Macrophage-selective iron loading induced by iron-dextran injection led to a significant increase in hepatic expressions of these factors in the NASH model, whereas the iron chelator 2,2’-bipyridyl treatment attenuated fibrogenesis. In this study, we will further investigate the molecular link between macrophage iron content and phenotypes during the development of NASH.
Major publications
M. Kawakubo, *M. Tanaka, K. Ochi, A. Watanabe, M. Saka-Tanaka, Y. Kanamori1, N. Yoshioka, S. Yamashita, M. Goto, M. Itoh, I. Shirakawa, S. Kanai, H. Suzuki, M. Sawada, A. Ito, M. Ishigami, M. Fujishiro, H. Arima, Y. Ogawa, *T. Suganami
“Dipeptidyl peptidase-4 inhibition prevents nonalcoholic steatohepatitis–associated liver fibrosis and tumor development in mice independently of its anti-diabetic effects”
Sci. Rep. 2020, 10, 983
doi: 10.1038/s41598-020-57935-6
M. Asakawa, *M. Itoh, *T. Suganami, T. Sakai, S. Kanai, I. Shirakawa, X. Yuan, T. Hatayama, S. Shimada, Y. Akiyama, K. Fujiu, Y. Inagaki, I. Manabe, S. Yamaoka, T. Yamada, S. Tanaka, *Y. Ogawa
“Upregulation of cancer-associated gene expression in activated fibroblasts in a mouse model of non-alcoholic steatohepatitis”
Sci. Rep. 2019, 9, 19601
doi: 10.1038/s41598-019-56039-0
M. Itoh, *T. Suganami, H. Kato, S. Kanai, I. Shirakawa, T. Sakai, T. Goto, M. Asakawa, I. Hidaka, H. Sakugawa, K. Ohnishi, Y. Komohara, K. Asano, I. Sakaida, M. Tanaka, *Y. Ogawa
“CD11c-positive resident macrophages drive hepatocyte death-triggered liver fibrosis in a murine model of non-alcoholic steatohepatitis”
JCI Insight 2017, 2, e92902
doi: 10.1172/jci.insight.92902
M. Tanaka, K. Ikeda, *T. Suganami, C. Komiya, K. Ochi, I. Shirakawa, M. Hamaguchi, S. Nishimura, I. Manabe, T. Matsuda, K. Kimura, H. Inoue, Y. Inagaki, S. Aoe, S. Yamasaki, *Y. Ogawa
“Macrophage-inducible C-type lectin underlies obesity-induced adipose tissue fibrosis”
Nat. Commun. 2014, 5, 4982
doi: 10.1038/ncomms5982
M. Itoh, *T. Suganami, N. Nakagawa, M. Tanaka, Y. Yamamoto, Y. Kamei, S. Terai, I. Sakaida, *Y. Ogawa
“Melanocortin-4 receptor-deficient mice as a novel mouse model of non-alcoholic steatohepatitis”
Am. J. Pathol. 2011, 179, 2454-2463
doi: 10.1016/j.ajpath.2011.07.014

