A03:Elucidation of the mechanisms of zinc homeostasis in the early secretory pathway and activation of zinc enzymes
Yuta AMAGAI
(IMRAM, Tohoku University)
Lab website

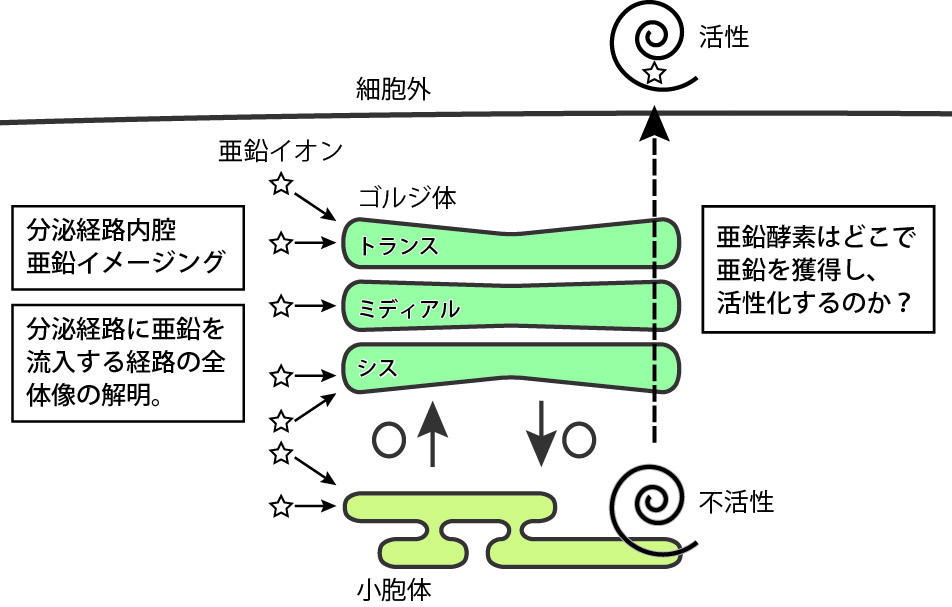
Early secretory pathway, which is composed of the endoplasmic reticulum and the Golgi apparatus, is a field of formation of native structures of newly synthesized secretory proteins. In the early secretory pathway, the qualities of proteins are strictly surveyed. We recently revealed that the function of ERp44, a member of PDI family, is enhanced by zinc ions at the lumen of the Golgi, and proposed that zinc regulates protein quality control in the early secretory pathway. Several members of ZnT/ZIP zinc transporters localize at the membranes of early secretory pathway, suggesting that zinc ions are fine-tuned spatiotemporally at these organelles. However, the most of mechanisms of zinc homeostasis at the early secretory pathway remain to be elusive. To address this question, we recently developed a new zinc probe and succeeded in a visualization of zinc dynamics in the Golgi apparatus. The goal of this project is to reveal the molecular mechanisms of zinc homeostasis at the early secretory pathway by using this probe and understand how zinc enzymes including ERp44 acquire zinc ions and mature through these organelles.
Major publications
S. Watanabe#, Y. Amagai#, S. Sannino#, T. Tempio, T. Anelli, M. Harayama, S. Masui, I. Sorrentino, M. Yamada, R. Sitia*, K. Inaba* (#; contributed equally, *; co-corresponding authors)
“Zinc regulates ERp44-dependent protein quality control in the early secretory pathway”
Nature Communications 2019, 10, 603
doi: 10.1038/s41467-019-08429-1
K. Arai#, T. Takei#, M. Okumura#, S. Watanabe#, Y. Amagai, Y. Asahina, L. Moroder, H. Hojo*, K. Inaba*, M. Iwaoka* (#; equally contributed, *; co-corresponding authors)
“Preparation of selenoinsulin as a long-lasting insulin analog”
Angewandte Chemie International Edition 2017, 56, 5522-5526
doi: 10.1002/anie.201701654
K. Maegawa, S. Watanabe#, K. Noi#, M. Okumura#, Y. Amagai#, M. Inoue, R. Ushioda, K. Nagata, T. Ogura, K. Inaba (#; contributed equally)
“The highly dynamic nature of ERdj5 is key to efficient elimination of aberrant protein oligomers through ER-associated degradation”
Structure 2017, 25, 846-857
doi: 10.1016/j.str.2017.04.001

