A01:Elucidation of the molecular mechanism underlying selenoprotein P function in selenium dynamics
Shun-ichi TANAKA
(Graduate School of Life and Environmental Sciences, Kyoto Prefectural University)
Lab website /
Researchmap

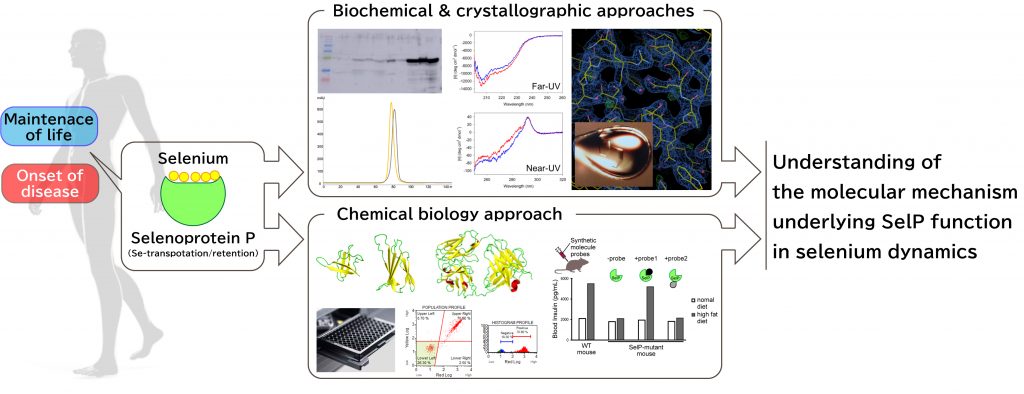
In nature, the essential trace element selenium exits predominantly as a selenocysteine residue (Sec) in living organisms. Sec-containing proteins (selenoproteins), such as well-known glutathione peroxidases and thioredoxine reductases, play important roles in human health, including roles in antioxidant defense, antitumor defense and other functions. Among the 25 selenoproteins so far identified in human, selenoprotein P (SelP) is the only one that has multiple selenocysteines in the amino acid sequence, and has been suggested to be involved in selenium transportation and/or retention in the human body. Previous and recent studies on SelP have shown that the protein function is intimately related to both maintenance of life and onset of disease, but the mechanistic details of these are poorly understood. In this project, we aim to elucidate the molecular mechanism underlying SelP function in selenium dynamics by employing an array of biochemical and crystallographic approaches as well as a chemical biology approach using synthetic binding proteins.
Major publications
S.-i. Tanaka, T. Takahashi, A. Koide, R. Iwamoto, S. Koikeda, S. Koide
“Monobody-mediated alteration of lipase substrate specificity”
ACS Chemical Biology 2018, 13, 1487-1492
doi: 10.1021/acschembio.8b00384
S.-i. Tanaka, T. Takahashi, A. Koide, S. Ishihara, S. Koikeda, S. Koide
“Monobody-mediated alteration of enzyme specificity”
Nature Chemical Biology 2015, 11, 762-764
doi: 10.1038/nchembio.1896
A. Sato, S, Yokotani, T. Tadokoro, S.-i. Tanaka, C. Angkawidjaja, Y. Koga, K. Takano, S. Kanaya
“Crystal structure of stable protein CutA1 from psychrotrophic bacterium Shewanella sp. SIB1”
Journal of Synchrotron Radiation 2011, 18, 6-10
doi: 10.1107/S0909049510028669
S.-i. Tanaka, H. Matsumura, Y. Koga, K. Takano, S. Kanaya
“Identification of the interactions critical for propeptide-catalyzed folding of Tk-subtilisin”
Journal of Molecular Biology 2009, 394, 306-319
doi: 10.1016/j.jmb.2009.09.028
S.-i. Tanaka, K. Saito, H. Chon, H. Matsumura, Y. Koga, K. Takano, S. Kanaya
“Crystal structure of unautoprocessed precursor of subtilisin from a hyperthermophilic archaeon: evidence for Ca2+-induced folding”
Journal of Biological Chemistry 2007, 282, 8246-8255
doi: 10.1074/jbc.M610137200

