A01:Molybdenum homeostasis by molybdenum storage protein
Nobutaka FUJIEDA
(Graduate School of Life and Environmental Sciences, Osaka Prefecture University)
Lab website

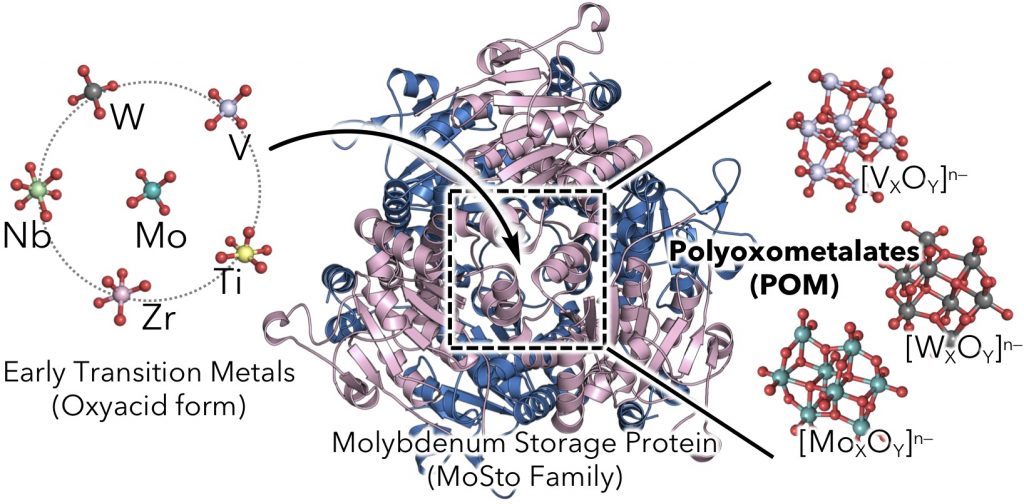
Molybdenum, tungsten, and vanadium play pivotal roles in the catalytic site of various enzymes. Generally, they exist in oxoacid form as molybdate, tungstate, and vanadate in water, not in cation like late transition metals, such as iron, copper, and zinc. Since they tend to form the polyoxometalate, their properties are different from those of sulfate and phosphate, although their molecular structures in water are very similar to these oxoacids of typical elements. Thereby, homeostasises of these early transition metals are still unknown in compared to those of late transition metals and oxoacid of typical elements. In this study, we tackle to unravel the uptake and sensing mechanism of the early transition metals by molybdenum storage protein family by using X-ray crystallography and spectroscopy. Furthermore, we discuss about “Integrated Bio-metal Science” from viewpoint of biological coordination chemistry including the integration of polyoxometalates.
Major publications
N. Fujieda*, H. Ichihashi, M. Yuasa, Y. Nishikawa, G. Kurisu, and S. Itoh*
“Cupin Variants as Macromolecular Ligand Library for Stereoselective Michael Addition of Nitroalkanes”
Angew. Chem. Int. Ed., 2020, in press
doi: 10.1002/anie.202000129
N. Fujieda*, T. Nakano, Y. Taniguchi, H. Ichihashi, H. Sugimoto, Y. Morimoto, Y. Nishikawa, G. Kurisu, and S. Itoh*
“A Well-Defined Osmium-Cupin Complex: Hyperstable Artificial Osmium Peroxygenase”
J. Am. Chem. Soc., 2017, 139, 5149-5155
doi: 10.1021/jacs.7b00675
N. Fujieda*, J. Schätti, E. Stuttfeld, K. Ohkubo, T. Maier, and T. R. Ward*
“Enzyme Repurposing of a Hydrolase as an Emergent Peroxidase upon Metal Binding”
Chem. Sci., 2015, 6, 4060-4065
doi: 10.1039/C5SC01065A
N. Fujieda, S. Yabuta, T. Ikeda, T. Oyama, N. Muraki, G. Kurisu*, and S. Itoh*
“Crystal Structures of Copper-Depleted and Copper-Bound Fungal Pro-Tyrosinase: Insights Into Endogenous Cysteine-Dependent Copper Incorporation”
J. Biol. Chem., 2013, 288, 22128-22140
doi: 10.1074/jbc.M113.477612
N. Fujieda, T. Ikeda, M. Murata, S.Yanagisawa, S. Aono, K. Ohkubo, S. Nagao, T. Ogura, S. Hirota, S. Fukuzumi, Y. Nakamura, Y. Hata, and S. Itoh*
“Post-translational His-Cys Cross Linkage Formation in Tyrosinase Induced by Copper(II)-Peroxo Species.”
J. Am. Chem. Soc., 2011, 133, 1180-1183
doi: 10.1021/ja108280w

